Draw the Electron Configuration for a Neutral Atom of Manganese
Doc Brown's Advanced Level Chemistry - Inorganic Chemistry - Periodic Table Revision Notes
Part 2 Electronic structure, spectroscopy & ionisation energies,
e lectron configurations of the elements for Z = 1 to 58
Section 2.3 Electron configurations for elements of atomic number Z = 1 to 56
Parts 2.1 to 2.2 should be read and studied first !
email doc brown - comments - query?
All my periodic table advanced A level chemistry revision notes
All my advanced A level inorganic chemistry revision study notes
GCSE Level periodic table revision notes
Use your mobile phone or ipad etc. in 'landscape' style
This is a BIG website, you need to take time to explore it [ SEARCH BOX ]
Sub-index for this page
Part 2.3 uses the rules on assigning electron arrangements, and how the quantum level notation is written out, and using boxes to represent orbitals, as well as the usual written orbital notation, is given for elements Z = 1 to 56.
e.g. 26 Iron, Fe
1s22s22p63s23p63d64s2
[Ar]3d
4s
4p

Part 2.4 The relationship between electron configuration and the Periodic Table and uses the electron configurations to show how the Periodic Table arises, i.e. an element's position in the Periodic Table and hence its chemistry, is primarily determined by the arrangement of its outer valency electrons.
Part 2.5 shows how to work out the electron configuration of ions, (positive cations or negative anions formed by the loss or gain of valence electrons) and relating electron arrangements to the oxidation states exhibited by selected elements.
See also on other pages part ...
2.1 The electronic basis of the modern Periodic Table
2.2 The electronic structure of atoms (including s p d f subshells/orbitals/notation)
2.6 Spectroscopy and the hydrogen spectrum
2.7 Evidence of quantum levels from ionisation energies
2.8 Emission and absorption spectra of elements
2.3 List of the Electronic Configuration of Elements 1 to 56 using the advanced notation
YOU MUST STUDY Parts 2.1 and 2.2 before studying section 2.3 onwards � The rules of how to assign electrons in multi�electron atoms to the appropriate quantum levels were explained in section 2.2 .
The list below quotes the ground state electron configurations i.e. the lowest available state according to the Aufbau principle (previously described). The order of filling the electron levels is listed below and also indicated on the diagram below.
Electron Box diagrams of the outer electron arrangement and examples of the simple electron notation (e.g. 2.8.1) are also included, with brief comments in the end right hand column e.g. element symbol, group, series etc.
The electrons�in�boxes notation for subshells: Boxes are used to represent an individual orbital or set of orbitals in the electrons are shown as arrows. The pairs up/down arrows represent a full orbital with electrons of opposite spin and note how the half�filled boxes/orbitals illustrate Hund's rule of maximum multiplicity
Energy level filling order to Z = 56 is 1s 2s 2p 3s 3p 4s 3d 4p (for Z = 1 to 36) then 5s 4d 5p 6s 4f/5d varies (for Z = 37 to 56)
However, when writing out the electron configuration you must write them out in order of strict principal quantum with the accompanying s, p, d, f notation
order of writing out is: 1s 2s 2p 3s 3p 3d 4s 4p 4d 4f 5s 5p 5d 6s (up to Z = 58)
BUT the order of orbital filling is: 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s (up to Z = 56)
So at Z = 21 Sc, you start to fill the 3d orbitals (NOT at Z = 20 Ca, and NOT 4p orbitals),
then at Z = 31 Ga you start to fill the 4p orbitals, and this is all you need pre-university!
| Atomic number Z and the element name and chemical symbol | Electron configuration Electron arrangement s, p, d & f notation with electron number superscripts (plus some simplified electron arrangements) | Electron spin box diagrams of the outer electron orbitals for the electron configuration of the atom representing the superscripted electrons beyond the inner noble gas core [He/Ne/Ar/Kr], the latter are not involved in chemical bonding/ or reactions. | Symbol, group/series/block and Comments Gp = Group! |
| 1 Hydrogen, H | 1s1 | 1s | H, no Gp really, a bit unique! |
| 2 Helium, He | 1s2= [He] | 1s | He, Gp 0/18 Noble Gas, |
| 3 Lithium, Li | 1s22s1 (simple notation: 2.1) | [He]2s | Li, s�block, Gp1 Alkali Metal, v. reactive |
| 4 Beryllium, Be | 1s22s2 (2.2) | [He]2s | Be, s�block, Gp2 Alkaline Earth Metal, |
| 5 Boron, B | 1s22s22p1 (2.3) | [He]2s | B, p�block, Gp3/13 |
| 6 Carbon, C | 1s22s22p2 (2.4) | [He]2s | C, p�block, Gp4/14, |
| 7 Nitrogen, N | 1s22s22p3 (2.5) | [He]2s | N, p�block, Gp5/15, |
| 8 Oxygen, O | 1s22s22p4 (2.6) | [He]2s | O, p�block, Gp6/16, |
| 9 Fluorine, F | 1s22s22p5 (2.7) | [He]2s | F, p�block, Gp7/17 Halogen, |
| 10 Neon, Ne | 1s22s22p6= [Ne] (2.8) | [He]2s | Ne, p�block, Gp 0/18 Noble Gas, |
| 11 Sodium, Na | 1s22s22p63s1 (2.8.1) | [Ne]3s | Na, Gp1 Alkali Metal, v. reactive |
| 12 Magnesium, Mg | 1s22s22p63s2 (2.8.2) | [Ne]3s | Mg, s�block, Gp2 Alkaline Earth Metal, |
| 13 Aluminium, Al | 1s22s22p63s23p1 (2.8.3) | [Ne]3s | Al, p�block, Gp3/13, |
| 14 Silicon, Si | 1s22s22p63s23p2 (2.8.4) | [Ne]3s | Si, p�block, Gp4/14, |
| 15 Phosphorus, P | 1s22s22p63s23p3 (2.8.5) | [Ne]3s | P, p�block, Gp5/15, |
| 16 Sulfur, S | 1s22s22p63s23p4 (2.8.6) | [Ne]3s | S, p�block, Gp6/16, |
| 17 Chlorine, Cl | 1s22s22p63s23p5 (2.8.7) | [Ne]3s | Cl, p�block, Gp7/17 Halogen, |
| 18 Argon, Ar | 1s22s22p63s23p6 = [Ar] (2.8.8) | [Ne]3s | Ar, p�block, Gp 0/18 Noble Gas, |
| 19 Potassium, K | 1s22s22p63s23p64s1 (2.8.8.1) | [Ar]3d | K, s�block, Gp1 Alkali Metal, v. reactive |
| 20 Calcium, Ca | 1s22s22p63s23p64s2 (2.8.8.1) | [Ar]3d | Ca, s�block, Gp2 Alkaline Earth Metal, |
| 21 Scandium, Sc | 1s22s22p63s23p63d14s2 | [Ar]3d | Sc, 3d block, not true Transition Metal |
| 22 Titanium, Ti | 1s22s22p63s23p63d24s2 | [Ar]3d | Ti, 3d block, a true Transition Metal |
| 23 Vanadium, V | 1s22s22p63s23p63d34s2 | [Ar]3d | V, 3d block, a true Transition Metal |
| 24 Chromium, Cr | 1s22s22p63s23p63d54s1 | [Ar]3d | Cr, 3d block, a true Transition Metal |
| 25 Manganese, Mn | 1s22s22p63s23p63d54s2 | [Ar]3d | Mn, 3d block, a true Transition Metal |
| 26 Iron, Fe | 1s22s22p63s23p63d64s2 | [Ar]3d | Fe, 3d block, a true Transition Metal |
| 27 Cobalt, Co | 1s22s22p63s23p63d74s2 | [Ar]3d | Co, 3d block, a true Transition Metal |
| 28 Nickel, Ni | 1s22s22p63s23p63d84s2 | [Ar]3d | Ni, 3d block, a true Transition Metal |
| 29 Copper, Cu | 1s22s22p63s23p63d104s1 | [Ar]3d | Cu, 3d block, a true Transition Metal |
| 30 Zinc, Zn | 1s22s22p63s23p63d104s2 | [Ar]3d | Zn, 3d block, not true Transition Metal |
| 31 Gallium, Ga | [Ar]3d104s24p1 | [Ar]3d | Ga, p�block, Gp3/13, |
| 32 Germanium, Ge | [Ar]3d104s24p2 | [Ar]3d | Ge, p�block, Gp4/14, |
| 33 Arsenic, As | [Ar]3d104s24p3 | [Ar]3d | As, p�block, Gp5/15, |
| 34 Selenium, Se | [Ar]3d104s24p4 | [Ar]3d | Se, p�block, Gp6/16, |
| 35 Bromine, Br | [Ar]3d104s24p5 | [Ar]3d | Br, p�block, Gp7/17 Halogen, |
| 36 Krypton, Kr | [Ar]3d104s24p6 = [Kr] (2.8.18.8) | [Ar]3d | Kr, p�block, Gp 0/18 Noble Gas, |
| 37 Rubidium, Rb | [Kr]5s1 | [Kr]5s | Rb, s�block, Gp1 Alkali Metal, v. reactive |
| 38 Strontium, Sr | [Kr]5s2 | [Kr]5s | Sr, s�block, Gp2 Alkaline Earth Metal, |
| 39 Yttrium, Y | [Kr]4d15s2 | [Kr]4d | Y, 4d block, not true Transition Metal |
| 40 Zirconium, Zr | [Kr]4d25s2 | [Kr]4d | Zr, 4d block, a true Transition Metal |
| 41 Niobium, Nb | [Kr]4d45s1 | [Kr]4d | Nb, 4d block, a true Transition Metal |
| 42 Molybdenum, Mo | [Kr]4d55s1 | [Kr]4d | Mo, 4d block, a true Transition Metal |
| 43 Technetium, Tc | [Kr]4d55s2 | [Kr]4d | Tc, 4d block, a true Transition Metal |
| 44 Ruthenium, Ru | [Kr]4d75s1 | [Kr]4d | Ru, 4d block, a true Transition Metal |
| 45 Rhodium, Rh | [Kr]4d85s1 | [Kr]4d | Rh, 4d block, a true Transition Metal |
| 46 Palladium, Pd | [Kr]4d10 | [Kr]4d | Pd, 4d block, a true Transition Metal |
| 47 Silver, Ag | [Kr]4d105s1 | [Kr]4d | Ag, 4d block, a true Transition Metal |
| 48 Cadmium, Cd | [Kr]4d105s2 | [Kr]4d | Cd, 4d block, not true Transition Metal |
| 49 Indium, In | [Kr]4d105s25p1 | [Kr]4d | In, p�block, Gp3/13, |
| 50 Tin, Sn | [Kr]4d105s25p2 | [Kr]4d | Sn, p�block, Gp4/14, |
| 51 Antimony, Sb | [Kr]4d105s25p3 | [Kr]4d | Sb, p�block, Gp5/14, |
| 52 Tellurium, Te | [Kr]4d105s25p4 | [Kr]4d | Te, p�block, Gp6/16, |
| 53 Iodine, I | [Kr]4d105s25p5 | [Kr]4d | I, p�block, Gp7/17 Halogen, |
| 54 Xenon, Xe | [Kr]4d105s25p6 = [Xe] | [Kr]4d | Xe, p�block, Gp 0/18 Noble Gas, |
| 55 Caesium, Cs | [Xe]6s1 | [Xe]6s | Cs, s�block, Gp1 Alkali Metal, v. reactive |
| 56 Barium, Ba | [Xe]6s2 | [Xe]6s | Ba, s�block, Gp2 Alkaline Earth Metal, |
| 57 Lanthanum, La | [Xe]5d16s2 | [Xe]5d | La, start of 5d�bock and Lanthanide Series |
| 58 Cerium, Ce | [Xe]4f26s2 not 4f15d16s2 | things get a bit less systematic in the f blocks | Ce, 1st of f�block in the Lanthanides Metals |
| ************************ | **************************************** | ********************************************************* | ***************************************************** |
The electron spin box diagrams can be used to show the full electronic structure e.g.





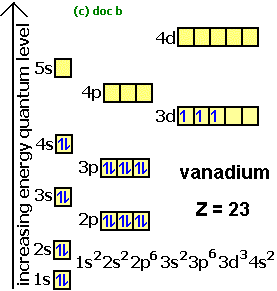
All based on the right-hand diagram
More 'quantum level quirks'!
A note on two anomalies in the 3d block , namely the transition metals chromium and copper:
Cr is [Ar]3d54s 1 and not [Ar]3d44s2
and Cu is[Ar]3d104s 1 and not [Ar]3d94s2
because an inner half�filled or fully�filled 3d sub�shells seem to be a little lower in energy level, and marginally more stable.
TOP OF PAGE and sub-indexes OR [Use the website search box]
2.4 Electron configuration and the Periodic Table
Not all the elements are shown but the position of s, p, d and f blocks are shown and explained after the table
It is an element's electron configuration that determines whether it is an s, p, d or f block element.
Instead of 'simply' assigning an element to its group or series because of its chemical properties e.g. formula of similar compounds, we can now assign an element's position in the periodic table using its electron configuration .
An s block element has one or two outer electrons i.e. ... s1 or ... s2 e.g. group 1 and group 2 metals (shown below)
A p block element has 1-6 outer p electrons beyond the s2 orbital i.e. ... s2p1 to s2p6 e.g. group 3/13 across to group 0/18 (shown below)
A d block element has 1 to 10 electrons in the outermost set of d orbitals i.e. ...d1 to ...d10 e.g. 3d block of Sc to Zn, 4d block Y to Cd (shown below)
Note that a true transition element has an ion with an incomplete d sub-shell (3d, 4d etc.)
An f block element has 1 to 14 electrons in the outermost set of f orbitals i.e. ...f1 to ...f14 e.g. Ce to Lu (NOT shown below)
This partial periodic table relates an element's electron configuration to the element's position in the periodic table.
You can then see the patterns between an atom's electron arrangement and the group, block or series the element belongs to.
| Pd | s block | 3d/4d blocks of Transition Metals (Periods 4/5), the 1st/10th are NOT true transition elements, they have no partially filled d shell in an ion. | p block elements | |||||||||||||||
| Gp1 | Gp2 | Gp3/13 | Gp4/14 | Gp5/15 | Gp6/16 | Gp7/17 | Gp0/18 | |||||||||||
| 1 | 1 H 1s1 | 2 He 1s2 | ||||||||||||||||
| 2 | 3 Li [He]2s 1 | 4 Be [He]2s2 | The e lectronic structure of Elements 1 to 56, ZSymbol, Z = atomic or proton number = total electrons in neutral atom, [He] = 1s2, [Ne] = 1s22s22p6, [Ar] = 1s22s22p63s23p6, [Kr] = 1s22s22p63s23p63d104s24p 6 Between Groups 2 and 3/13 are the d�blocks and f�blocks where the quantum energy level rules permit their inclusion. Periods 4 and 5 have 18 elements each, including the 3d (Sc-Zn) and 4d (Y-Cd) d blocks of elements respectively (Groups 3 to 12 � new notation). | 5 B [He]2s22p1 | 6 C [He]2s22p2 | 7 N [He]2s22p3 | 8 O [He]2s22p4 | 9 F [He]2s22p5 | 10 Ne [He]2s22p6 | |||||||||
| 3 | 11 Na [Ne]3s1 | 12 Mg [Ne]3s2 | 13 Al [Ne]3s23p1 | 14 Si [Ne]3s23p2 | 15 P [Ne]3s23p3 | 16 S [Ne]3s23p4 | 17 Cl [Ne]3s23p5 | 18 Ar [Ne]3s23p6 | ||||||||||
| 4 | 19 K [Ar]4s1 | 20 Ca [Ar]4s2 | 21 Sc [Ar] 3d14s2 | 22 Ti [Ar] 3d24s2 | 23 V [Ar] 3d34s2 | 24 Cr [Ar] 3d54s1 | 25 Mn [Ar] 3d54s2 | 26 Fe [Ar] 3d64s2 | 27 Co [Ar] 3d74s2 | 28 Ni [Ar] 3d84s2 | 29 Cu [Ar] 3d104s1 | 30 Zn [Ar] 3d104s2 | 31 Ga [Ar] 3d104s24p1 | 32 Ge [Ar] 3d104s24p2 | 33 As [Ar] 3d104s24p3 | 34 Se [Ar] 3d104s24p4 | 35 Br [Ar] 3d104s24p5 | 36 Kr [Ar] 3d104s24p6 |
| 5 | 37 Rb [Kr]5s 1 | 38 Sr [Kr]5s2 | 39 Y [Kr] 4d15s 2 | 40 Zr [Kr] 4d25s2 | 41 Nb [Kr] 4d45s1 | 42 Mo [Kr] 4d55s1 | 43 Tc [Kr] 4d55s2 | 44 Ru [Kr] 4d75s1 | 45 Rh [Kr] 4d85s1 | 46 Pd [Kr] 4d10 | 47 Ag [Kr] 4d105s1 | 48 Cd [Kr] 4d105s2 | 49 In [Kr] 4d105s25p1 | 50 Sn [Kr] 4d105s25p2 | 51 Sb [Kr] 4d105s25p3 | 52 Te [Kr] 4d105s25p4 | 53 I [Kr] 4d105s25p5 | 54 Xe [Kr] 4d105s25p6 |
| 6 | 55 Cs [Xe]6s 1 | 56 Ba [Xe]6s 2 | 4f�block (14) and 5d�block (10), total of 32 elements in period 6 including the Lanthanide Series of Metals. | 81 Tl [Xe] 4f145d106s26p1 | 82 Pb [Xe] 4f145d106s26p2 | 83 Bi [Xe] 4f145d106s26p3 | 84 Po [Xe] 4f145d106s26p4 | 85 At [Xe] 4f145d106s26p 5 | 86 Rn [Xe] 4f145d106s26p 6 | |||||||||
| 7 | 87 Fr [Rn]7s1 | 88 Ra [Rn]7s 2 | 5f�block and 6d�block including the Actinide Series of Metals in period 7, see full table below | 113 Nh [Rn] 5f146d107s27p1 | 114 Fl [Rn] 5f146d107s27p2 | 115 Mc [Rn] 5f146d107s27p3 | 116 Lv [Rn] 5f146d107s27p4 | 117 Ts [Rn] 5f146d107s27p5 | 118 Og [Rn] 5f146d107s27p6 | |||||||||
-
Note on Group numbers
-
Using 0 to denote the Group number of Noble Gases is very historic now since compounds of xenon known exhibiting a valency of 8.
-
Because of the horizontal series of elements e.g. like the Sc to Zn block (10 elements), Groups 3 to 0 can also be numbered as Groups 13 to 18 to fit in with the actual number of vertical columns of elements and this is the modern trend in periodic table notation.
-
This can make things confusing, but there it is, classification is still in progress!
-
The atomic/proton number, decides which element an atom is and the outer electron structure decides which group/block/series the element belongs to and ultimately its chemistry.
-
The positions of the s, p, d and f blocks are also indicated Periodic Table above and arise from the quantum rules
-
s block elements have an outer shell of just 1 or 2 s electrons i.e. s1 or s2 configuration beyond an inner noble gas configuration (2 per period from period 2 onwards), that is groups 1 and 2. Technically, hydrogen and helium are in the s block, but bare little chemical similarity with the group 1 and group 2 metals.
-
p block elements have an outer electron configuration of s2p1 to s2p6 i.e. elements where the p sub-shell is being filled (6 per period from period 2 onwards), that is groups 3/13 to 0/18.
-
d block elements eg the 3d (Sc-Zn) where the 3d sub-shell is being filled and like wise for the 4d block (Y-Cd),10 elements per block per period from period 4 onwards, the first horizontal blocks of metals which lie between the s block and p block,
-
f blocks elements eg the 4f and 5f blocks where the f sub-shells start being filled (14 elements in each block per period from period 6 onwards).
-
-
-
The most stable electron configurations
-
When the outer s and p quantum levels (and any completely filled inner orbital quantum levels eg 3d or 4f) you get a particularly stable element with minimal chemical reactivity ie you get a Noble Gas element [simple electron notation in ()]
-
Z = 2, helium, 1s2= [He] (2)
-
Z = 10, neon, 1s22s22p6= [Ne] (2.8)
-
Z = 18, argon, 1s22s22p63s23p6 = [Ar] (2.8.8)
-
Z = 36, krypton, [Ar]3d104s24p6 = [Kr] (2.8.18.8)
-
Z = 54, xenon, [Kr]4d105s25p6 = [Xe]
-
Z = 86, radon, [Xe]5d106s26p6 =[Rn]
-
-
-
What is the electronic basis of Groups of elements? � their 'electronic classification '
-
For groups 1 to 2, and 'old' 3 to 0/'new' notation 13 to 18 (except He), all the elements in the same vertical column have the same outer electron configuration and therefore will be expected to have a very similar chemistry.
-
This gives the electronic basis for Mendeleev's brilliant conception of the periodic table, ie laying out all the elements in order of 'atomic weight' and lining them up to give vertical columns of chemically and physically similar elements.
-
-
For the d blocks of Groups 3 to 12, using the 'new' group number notation, the vertical 'group' connection of similar outer electron configuration is consistent except for V/Nb, Fe/Ru, Co/Rh, Ni/Pd where the 3d/4s and 4d/5s pairs of levels are of very similar energy and small differences in outer electron configuration occur.
-
Never�the�less these pairs of elements show strong similarities as part of the justification for denoting the Transition Metals plus Groups 4 to 0 as Groups 3 to 18.
-
-
-
What is the electronic basis for the 'series of elements'? � their 'electronic classification '
-
The '1st Transition Metals Series' from Sc to Zn, and other 'horizontal blocks' are sometimes called a 'series' but they are better described as the '3d block' or '3d series of elements' (and, 4d block, 4f block � filling of 4f sub�shell etc.), but a horizontal row of elements, unlike the vertical columns of the eight vertical groups.
-
Why is 'block' better than 'series'?
-
The reference to the electronic structure is very important, the word series is a bit vague!
-
Technically, scandium (Sc, Z = 21) and zinc (Zn, Z = 30), are NOT true transition metals BUT they are true 3d block elements!
-
See Detailed Advanced Level Transition Metal Notes
-
-
-
-
-
What is the overall electronic basis for blocks of elements across the whole of the periodic table?
-
The s�block consists of Groups 1 and 2 where the only outer electrons are in an s sub�energy level orbital (no outer p electrons, 2 per period).
-
Technically, the same applies to hydrogen and helium in period 1.
-
-
The p�block corresponds to Groups 3 to 0 (old notation) or Groups 13 to 18 (new notation) where the three p sub�energy level orbitals are being filled (6 per period).
-
Starting with period 4, where the first of the d sub�shells is low enough in energy to be filled, there are ten elements 'inserted' between groups 2 and 3, the so�called d blocks of ten elements (the 1st block, the 3d block Sc�Zn is on Period 4).
-
Therefore Sc to Zn form the head elements of Groups 3 to 12 using the 'new' group number notation.
-
Similarly on period 5 there is a 4d block where the 4d sub�shell level is filled.
-
So 10 d block elements per period are now permitted\under the quantum number rules.
-
-
Starting with cerium (Z=58, period 6), see in full table below, there is a further insertion of fourteen elements where the seven f�orbital sub�shell is being filled after the first of the d�block metals and similarly with thorium (Z=90) in period 7 and these are known as the f blocks (14 per period where permitted).
-
-
The full Periodic Table is shown below without the electron configurations, but including the old/new group number notation.
| Pd | s�block metals | 3d to 6d blocks including the Transition Metals For Periods 4 to 7, note that the 1st (d1) and 10th (d10) block metals are NOT true transition elements, the d2 to d9 elements are true transition metals | p�block metals and non-metals | |||||||||||||||
| Gp1 | Gp2 | Gp3/ 13 | Gp4/ 14 | Gp5/ 15 | Gp6/ 16 | Gp7/ 17 | Gp0/ 18 | |||||||||||
| 1 | 1H Note: (i) H does not readily fit into any group, (ii) He not strictly a 'p' element but does belong in Gp 0/18 | 2He | ||||||||||||||||
| 2 | 3Li | 4Be | Full IUPAC modern Periodic Table of Elements Z Symbol, z = atomic or proton number | 5B | 6C | 7N | 8O | 9F | 10Ne | |||||||||
| 3 | 11Na | 12Mg | *Gp3 | *Gp4 | *Gp5 | *Gp6 | *Gp7 | *Gp8 | *Gp9 | *Gp10 | *Gp11 | *Gp12 | 13Al | 14Si | 15P | 16S | 17Cl | 18Ar |
| 4 | 19K | 20Ca | 21Sc | 22Ti | 23V | 24Cr | 25Mn | 26Fe | 27Co | 28Ni | 29Cu | 30Zn | 31Ga | 32Ge | 33As | 34Se | 35Br | 36Kr |
| 5 | 37Rb | 38Sr | 39Y | 40Zr | 41Nb | 42Mo | 43Tc | 44Ru | 45Rh | 46Pd | 47Ag | 48Cd | 49In | 50Sn | 51Sb | 52Te | 53I | 54Xe |
| 6 | 55Cs | 56Ba | *57-71 | 72Hf | 73Ta | 74W | 75Re | 76Os | 77Ir | 78Pt | 79Au | 80Hg | 81Tl | 82Pb | 83Bi | 84Po | 85At | 86Rn |
| 7 | 87Fr | 88Ra | *89-103 | 104Rf | 105Db | 106Sg | 107Bh | 108Hs | 109Mt | 110Ds | 111Rg | 112Cn | 113Nh | 114Fl | 115Mc | 116Lv | 117Ts | 118Og |
| Group 1 Alkali Metals Group 2 Alkaline Earth Metals Group 7/17 Halogens Group 0/18 Noble Gases Take note of the four points on the right | ||||||||||||||||||
| * 57La | 58Ce | 59Pr | 60Nd | 61Pm | 62Sm | 63Eu | 64Gd | 65Tb | 66Dy | 67Ho | 68Er | 69Tm | 70Yb | 71Lu | ||||
| * 89Ac | 90Th | 91Pa | 92U | 93Np | 94Pu | 95Am | 96Cm | 97Bk | 98Cf | 99Es | 100Fm | 101Md | 102No | 103Lr | ||||
| *Horizontal insert in Period 6 of Lanthanide Metal Series (Lanthanides/Lanthanoids) Z=57 to 71 includes 4f�block series (elements 58�71). Element 57 is the start of the 5d block, interrupted by the 14 4f block elements and then continues with elements 72-80. *Horizontal insert in Period 7 of the Actinide Series of Metals (Actinides/Actinoids) Z=89�103 including the 5f�block series (elements 90�103). Element 57 is the start of the 5d block, interrupted by the 15 5f block elements and continues with elements 72-80. | ||||||||||||||||||
| ||||||||||||||||||
-
Notes:
-
The Noble Gases have been referred to as Group 0 because they were believed not to form compounds with other elements.
-
However, since 1961, many compounds of xenon have been prepared including xenon(VIII) oxide, XeO4, thus attaining the expected maximum possible oxidation state, so Group 18 seems most appropriate to use these days for advanced level courses.
-
-
The d block elements are sometimes referred in terms of vertical columns as Groups 3 to 12, and the subsequent p�block group columns as Groups 13 to 18.
-
Whether this will become standard at pre�university level, I don't know? but I think its a bit confusing for school level chemistry below pre�university courses like UK GCSE or US grades 6�10.
-
-
-
The s p d f blocks are shown in the Periodic Table above.
TOP OF PAGE and sub-indexes OR [Use the website search box]
2.5 Electronic configuration of ions and oxidation states
How do you work out the electron arrangement of ions? How do you work out the electron configuration of ions?
In what order to you remove electrons for positive ions? In what order do you add electrons for negative ions?
-
The electron configuration of ions :
-
Beware in quoting the configurations for simple ions where, although the order of removal is basically the reverse of the order for filling the energy levels, there is one important exception you should know.
-
For d/transition blocks/series the 4s electrons are 'removed' before the 3d electrons and similarly the 5s electrons are 'removed' before the 4d electrons.
-
-
Positive ions (cations) are formed by electron loss and the order of removal is the reverse of the order the full electron configuration is written out e.g.
-
sodium atom Na = 1s22s22p63s1 , sodium ion Na+ = 1s22s22p6 = [Ne]
-
calcium atom is Ca = 1s22s22p63s23p64s2, calcium ion Ca2+ = 1s22s22p63s23p6 = [Ar]
-
iron atom Fe = [Ar]3d64s2, iron(II) ion Fe2+ = [Ar]3d6 , and iron(III) ion Fe3+ = [Ar]3d5
-
germanium atom Ge = [Ar]3d104s24p2, germanium(II) ion Ge2+ = [Ar]3d104s2 , germanium(IV) ion Ge4+ = [Ar]3d10
-
-
Negative ions (anions) are formed by electron gain and the filling order rule is continued e.g.
-
chlorine Cl = [Ne]3s23p5, chloride ion Cl� = [Ne]3s23p6 = [Ar]
-
oxygen: O = [He]2s22p4, oxide ion O2� = [He]2s22p6 = [Ne]
-
phosphorus: P = [Ne]3s23p3, phosphide ion P3� = [Ne]3s23p3 = [Ar]
-
Note the maximum negative oxidation state is governed by the number of electrons needed to complete a noble gas structure in the covalent or ionic bonding situation.
-
-
-
Oxidation state and electronic structure :
-
For more details see notes on oxidation state and redox reactions .
-
The maximum oxidation state is often, but not always, limited by an inner full noble gas structure with or without a full d/f sub�shell.
-
The maximum oxidation state from Group 1 Alkali Metals (+1) to Group 0/18 Noble Gases (+8) is numerically equal to the number of outer electrons, i.e. those beyond an inner noble gas core or inner noble gas plus a full d/f sub�shell e.g.
-
The maximum oxidation states for Groups 1 to 0 in old notation (now s-block Groups 1-2 and p-block 13-18)
-
Group 1 , outer electron configuration s 1 , eg sodium is +1 in sodium chloride NaCl
-
Group 2 , outer electron configuration s 2 , eg magnesium is +2 in magnesium oxide MgO
-
Group 3 (13) , outer electron configuration s2p 1 , eg aluminium is +3 in aluminium fluoride AlF3
-
Group 4 (14) , outer electron configuration s2p 2 , eg silicon is +4 in silicon dioxide SiO2
-
Group 5 (15) , outer electron configuration s2p 3 , eg phosphorus is +5 in the phosphate(V) ion PO4 3�
-
Group 6 (16) , outer electron configuration s2p 4 , eg sulfur is +6 in sulfur trioxide SO3
-
Group 7 (17) , outer electron configuration s2p 5 , eg chlorine is +7 in the chlorate(VII) ion ClO4 �
-
Group 0 (18) , outer electron configuration s2p 6 (except He , just s 2 ), eg xenon is +8 in xenon(VIII) oxide XeO4
-
The maximum oxidation state pattern for the d blocks is a bit more complicated and the trend goes through a maximum e.g.
-
the maximum observed oxidation states for the 3d block and transition metal series :
-
Sc (+3), Ti (+4), V (+5), Cr (+6), Mn (+7), Fe (+3, maybe +6?), Co (+3), Ni (+3), Cu (+3), Zn (+2)
-
The maximum oxidation state from scandium to manganese (Sc-Mn) equates to the presence of 4 to 7 3d and 4s electrons beyond the inner noble gas core of [Ar], the 'theoretical' ion would have a 3d0 structure.
-
-
-
-
Atoms or ions with the same electron configuration are referred to as isoelectronic eg
-
Na+, Mg2+, Al3+ and Ne are all 1s22s22p6 and you might say the three ions are isoelectronic with neon.
-
N3-, O2- and F- are also all isoelectronic with neon.
-
Zn2+ and Ge4+ are both [Ar]3d10
-
-
SPECTROSCOPY, the HYDROGEN SPECTRUM and IONISATION ENERGY PATTERNS
are now on a separate page
TOP OF PAGE and sub-indexes OR [Use the website search box] OR links below















Website content � Dr Phil Brown 2000+. All copyrights reserved on revision notes, images, quizzes, worksheets etc. Copying of website material is NOT permitted. Exam revision summaries & references to science course specifications are unofficial.
Source: https://docbrown.info/page07/ASA2ptable2a.htm
0 Response to "Draw the Electron Configuration for a Neutral Atom of Manganese"
Post a Comment